On September 8, 2025, the U.S. Department of Health & Human Services’ Office of Inspector General (HHS-OIG) issued a report expressing “major” fraud, waste, and abuse concerns related to skin substitutes billed to Medicare Part B.
According to the OIG’s report, Medicare Part B reimbursement for skin substitutes “skyrocketed” over the past 2 years, with nearly $3 billion paid through the first 3 quarters of 2024, and a total of over $10 billion over the past 2 years. This represents a 640% increase in Part B spending on skin substitutes since 2022. The report concludes that “the rapid growth in expenditures was driven by both increased utilization and higher prices.”
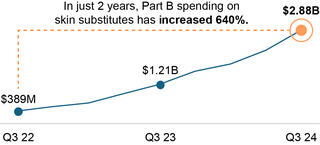
Chart from OIG Report
The OIG’s report lists the following aspects of Part B spending as raising “serious concerns”:
- Large increases in the number of enrollees with skin substitute claims and the amount of product billed for each enrollee, particularly in home care.
- A massive gap in spending between Part B and Medicare Advantage.
- A steep rise in the cost of individual skin substitutes combined with providers’ propensity to shift to more and more expensive products.
- Fraud schemes that allow bad actors to quickly get paid tens of millions of dollars when billing for just a small number of Part B enrollees.
The report attributes these alarming trends to (1) manufacturers’ ability to quickly bring new skin substitutes to the market compared to typical products paid using ASP, and (2) financial incentives that make certain products more attractive to providers. The OIG expressed concern that under the current payment system, Medicare often pays providers for skin substitutes in an amount that is much higher than the providers’ purchase price, allowing providers to keep the “spread,” which, according to the OIG, creates incentives for providers to bill for more and more units of skin substitutes and choose the products with the greatest spread.
The report also noted that home care represented a “disproportionate share” of Part B expenditures. According to the OIG, 28% of enrollees with a paid skin substitutes claim under Part B were being treated in their home as opposed to in an office setting. Moreover, the report states that home care providers billed for substantially more units and “markedly” more expensive products than office-based providers.
The report contains a “call to action,” noting that “action is urgently needed to rein in the massive increases in Medicare Part B spending for skin substitutes. Specifically, “OIG’s findings illustrate the critical need for payment reforms that address fraud, waste, and abuse in Medicare skin substitute billing.” In addition to asking policymakers to consider legislative options, the OIG’s report lists steps that CMS has already taken to address these concerns including (1) a CY2026 physician fee schedule proposed rule that would change the Part B payment methodology for skin substitutes, and (2) a 6-year model “that uses utilization management and artificial intelligence technologies to implement and streamline prior authorization for potentially fraudulent, wasteful, or harmful high-cost services in Medicare Part B.”
_________________________
The attorneys at Chilivis Grubman represent healthcare providers in connection with payor audits and audit appeals, government investigations, and other regulatory, compliance, and litigation matters. If you need assistance with such a matter, contact us today.
